Tonner One World Holdings (OTC:TONR ) About to Explode?
Company Name: Tonner-One World Holdings, Inc.
Ticker: TONR
Exchange: OTC
Website: https://twitter.com/TonnerOne
Company Summary: Tonner-One World Holdings, Inc is an actively expanding Fintech company seeking acquisitions with an emphasis on Crypto and Crypto Exchanges with the most potential for future growth.
Why did it go up over 100%?
November 18th Tonner announces “As a shareholder of Tonner-One World Holdings, Inc., you are receiving this letter to inform you of a recent decision by your company’s Board of Directors to transform its business model to precisely focus on acquisitions and operation of businesses and assets in the fast-growth Fintec growth industry.”
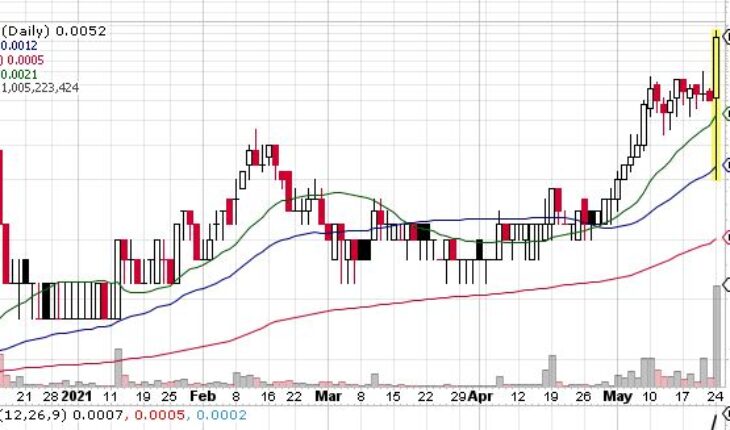
5 Day Technical Analysis:
The stock has been gapping up premarket all week, so if the stock can gap up in the morning and beat $.0096 tomorrow, I like it. However because of the lack of news and the 80% run of late, be very careful.
1 Day Technical Analysis
Volume is stable and it is consolidating with a slight bearish trend within the consolidation on the 1 day chart. Again, if it can beat $.0096 I like it!
BELLUS Health Inc. (Nasdaq:BLU; TSX:BLU) announces 2b clinical trials!
Company Name: BELLUS Health Inc.
Ticker: (BLU)
Exchange: NASDAQ
Website: www.bellushealth.com
Company Summary:
BELLUS Health is a clinical-stage biopharmaceutical company developing novel therapeutics for the treatment of RCC and other hypersensitization-related disorders. The Company’s product candidate, BLU-5937, has successfully completed a Phase 2b trial in RCC and is planning a Phase 3 program that is expected to begin in 2022.
Why did it go up almost 100%?
Link HERE – LAVAL, Quebec, December 13, 2021–(BUSINESS WIRE)–BELLUS Health Inc. (Nasdaq:BLU; TSX:BLU) (“BELLUS Health” or the “Company”), a clinical-stage biopharmaceutical company developing novel therapeutics for the treatment of refractory chronic cough (“RCC”) and other hypersensitization-related disorders, today announced that the 50 mg and 200 mg BID doses of BLU-5937 in its Phase 2b SOOTHE trial for the treatment of RCC achieved statistical significance on the primary endpoint with 34% placebo-adjusted reduction in 24-hour cough frequency observed (p ≤ 0.005) at day 28. BLU-5937 was generally well-tolerated, with low rates of taste-related adverse events reported (≤6.5%) at all doses.
Mr. Roberto Bellini, Chief Executive Officer of BELLUS Health commented, “We are extremely pleased with the compelling topline results from the Phase 2b SOOTHE trial which highlight BLU-5937’s best-in-class potential for the treatment of refractory chronic cough. RCC is a prevalent and growing condition that significantly impacts the quality of life of an estimated 9 million patients in the United States and 9 million patients in Europe.”
5 Day Technical Analysis:
There are a lot of traders at $9.05, so if this stock can beat $9.05 tomorrow, I like it. If not, and I’m thinking it won’t, stay away. Caveat, the news is really good, so I hold out hope, but I don’t like the huge 70% gap up. Happy trading and always play it safe with trailing stops!
Biofrontera Inc. (Nasdaq: BFRI Patent allowed!

Company Name: Biofrontera Inc.
Ticker: (BFRI)
Exchange: NASDAQ
Website: www.biofrontera-us.com
Company Summary:
Biofrontera Inc. is a U.S.-based biopharmaceutical company commercializing a portfolio of pharmaceutical products for the treatment of dermatological conditions with a focus on photodynamic therapy (PDT) and topical antibiotics. The Company’s licensed products are used for the treatment of actinic keratoses, which are pre-cancerous skin lesions, as well as impetigo, a bacterial skin infection.
Why did it go up over 100%?
WOBURN, Mass., Dec. 08, 2021 (GLOBE NEWSWIRE) — Biofrontera Inc. (Nasdaq: BFRI), a biopharmaceutical company specializing in the commercialization of dermatological products, announced today that the United States Patent and Trademark Office (USPTO) has issued a Notice of Allowance for Biofrontera Pharma GmbH’s U.S. patent number 17/215,785 (‘785 patent), titled “Illumination device for photodynamic therapy, method for treating a skin disease and method for operating an illumination device,” which protects a number of innovations relating to the RhodoLED XL lamp.
WOBURN, Mass., Dec. 13, 2021 (GLOBE NEWSWIRE) — Biofrontera Inc. (Nasdaq: BFRI), a biopharmaceutical company specializing in the commercialization of dermatological products, announced today that the first subject has been enrolled in the Phase 2b study performed by Biofrontera Bioscience GmbH to evaluate the safety and efficacy of Ameluz® in combination with the red-light lamp BF-RhodoLED® for the treatment of moderate-to-severe acne with photodynamic therapy (Ameluz®-PDT).
5 Day Chart Technical Analysis
I like the stock! Great news and the 5 day is bullish and has solid news with a patent being accepted and a phase 2b clinical trial being announced.
1 Day Chart Technical Analysis
Technical Analysis:
I like the stock only IF the bearish trend continues in the morning, stay AWAY until there is consolidation and a double confirmation of a bullish move.
Petros Pharma (PTPI) to launch an OTC Erectile dysfunction pill?

Company Name: Petros Pharmaceuticals, Inc.
Ticker: (PTPI)
Exchange: NASDAQ
Website: https://STENDRA.com/.
Company Summary:
Petros Pharmaceuticals is committed to the goal of becoming a world-leading specialized men’s health company by identifying, developing, acquiring, and commercializing innovative therapeutics for men’s health issues, including, but not limited to, erectile dysfunction, endothelial dysfunction, psychosexual and psychosocial ailments, Peyronie’s disease, hormone health, and substance use disorders.
Why did it go up over 100%?
Petros Pharmaceuticals, Inc. (“Petros” or “the Company”) (Nasdaq: PTPI), a leading provider of therapeutics for men’s health, today reports positive over-the-counter (OTC) draft label comprehension study results for its erectile dysfunction (ED) Drug STENDRA® (avanafil). This Pivotal Label Comprehension Study was designed to assess comprehension of a draft STENDRA® Drug Facts Label intended for OTC use. This study is a key component in the Company’s plans to engage in discussions with the FDA to expand the product’s access through application for a possible OTC pathway. Early study indicators show positive label comprehension outcomes in the study, which has encouraged the Company to initiate the next step, a Self-Selection Study, as it continues to build the case for the FDA for OTC access to STENDRA.
“The label comprehension study is a key component of our plans to help expand access for STENDRA beyond the prescription model, and to make STENDRA potentially the first prescription-grade ED medication to become available over-the-counter in the United States. While the process of gathering data to be shared with the U.S. Food and Drug Administration (FDA) continues, we are enthusiastic about our progress toward providing new hope for the millions of men suffering from this condition,” commented Fady Boctor, Petros’s President and Chief Commercial Officer.
The first “potentially the first prescription-grade ED medication to become available over-the-counter in the United States.” EVER?!?
I’m a big fan of this company and the recent news it released. Let’s take a look at the chart.
5 Day Technical Analysis:
It’s been gapping up and pulling back for the last 4 days and it looks like it is still bullish but it is possibly about to go bearish! Let’s tak a close look at the 1 day.
1 Day CHart Technical Analysis
It’s very simple, can it beat $4.38? Yes, then I would wait for a pull back and then swing trade it for a quick scalp. If it can’t, I would expect a pullback then consolidation and then a run! Overall, I love the stock because of the news. The first OTC ED med, say what!?!?
Tantech Holdings Ltd (NASDAQ: TANH) develops driverless street sweepers!

Company Name: Tantech Holdings Ltd
Ticker: (TANH)
Exchange: NASDAQ
Website: http://ir.tantech.cn
Company Summary:
For the past decade, Tantech has been a highly specialized high-tech enterprise producing, researching and developing bamboo charcoal-based products with an established domestic and international sales and distribution network. Since 2017, when the Company acquired 70% of Shangchi Automobile, a vehicle manufacturer based in Zhangjiagang City, Jiangsu Province, it has manufactured and sold vehicles. The Company established two new subsidiaries, Lishui Smart New Energy Automobile Co., Ltd. and Zhejiang Shangchi New Energy Automobile Co., Ltd., in November 2020, to produce and sell street sweepers and other electric vehicles.
Why did it go up over 100%?
LISHUI, China, Dec. 13, 2021 /PRNewswire/ — Tantech Holdings Ltd (NASDAQ: TANH) (“Tantech” or the “Company”), a clean energy company in China, today announced that its subsidiary, Lishui Smart New Energy Automobile Co., Ltd., received a milestone order for twelve of its highly innovative SC-120A driverless and autonomous street sweepers. The customer is a real estate development and property management company, managing industrial parks and other properties in China. This represents the first order since the recent completion of the final phase of factory testing and standard factory quality control reviews. The Company separately announced the successful development of its third generation of driverless and autonomous sweeper with enhanced features, including expanded route and memory capabilities, additional sensors and improved further improved navigation and positioning accuracy.
Our opinion:
Pretty cool, driverless street sweepers! Autonomous vehicles are going to be the norm in the future and they just sold 12, pretty big news!
1 Day
1 Day Technical Analysis:
I like it, the news is great with driverless street sweepers. The chart is solid, my only caveat is, wait until it breaks $.63 in my opinion. Hopefully it gaps up this morning and breaks through.
5 Day
5 Day Technical Analysis:
It’s a bullish trend line, hopefully the volume will continue. Keep an eye on the news feed, that drives volume. Again, the $.63 PPS is an important target for this morning and it would be nice to see it glide right by it. Happy trading!
