On March 4, 2026, COEP stock opened at...
Healthcare
ENDRA Life Sciences Is Solving a $2,500 Problem in a $50 Billion Market — and the Window May Be Opening
ENDRA Life Sciences Inc. (NASDAQ:...

Sponsored Alert: TMGI
Dates: 2/6/2026 Transglobal Management...
ACMB Stock Review
Dates: 2/3/2026 ...
Mangoceuticals (MGRX): A Low Float Stock on the Verge of a Short Squeeze?
In the fast-moving world of biotech...
Coeptis Therapeutics Could Revolutionize The Treatment of Cancer
Small Cap Exclusive is proud to...
NMRD Stock: Why is Nemaura Medical (NASDAQ: NMRD) A Top Stock To Watch in February?
Over the past week, the Nemaura Medical Inc (NASDAQ: NMRD) stock has been in the middle of a solid rally and recorded gains of 56% to emerging as one of the stock to watch. As it happens, the NMRD stock hit its highest level since mid-October last year.
Nemaura Medical Inc
Company Name: Nemaura Medical Inc
Ticker: NMRD
Exchange: NASDAQ
Website: https://nemauramedical.com

Nemaura Medical Inc Summary:
Nemaura Medical Inc. is a medical technology company developing and commercialising non-invasive wearable diagnostic devices. The Company is currently also commercialising sugarBEAT® and proBEAT™. sugarBEAT®, a CE mark approved Class IIb medical device, as a non-invasive and flexible continuous glucose monitor (CGM) providing actionable insights derived from real time glucose measurements and daily glucose trend data, which may help people with diabetes and prediabetes to better manage, reverse, and prevent the onset of diabetes.
There are certain specific triggers that led to the remarkable rally in the NMRD stock.

#1 Medical’s Program Shows Weight Loss In 100% Of Participants
One of those came about earlier in the week on January 24 when the company announced the initial results from the patient studies that had been conducted with the National Health Service (NHS) of the United Kingdom for Miboko, its metabolic health program.
The results were positive as the patients recorded an average weight loss of 3.7 pounds and all the patients experienced some degree of weight improvement.
This news had a major positive impact on NMRD stock and is one of the top stock to watch.

#2 Commences Pilot Program of Miboko with the NHS
While that was quite clearly a significant development it should be noted that the announcement with regards to the study for Miboko in collaboration with the NHS had been announced back on November 29, 2022.
Mikobo is the first product in its category to have a non-invasive glucose sensor in a lifestyle app. It not includes information with regard to drinking and good intake but also provides analytics and educational content. At the end of the analysis, the app provides the user with a metabolic score based on diet and other important factors.

#3 Solid Industry Growth
Nemaura is entering the Continuous Glucose Monitoring Market with the product and it is a market that is expanding rapidly. The market was worth as much as $6.13 billion back in 2021 and by 2030 it is expected to command a valuation of $16.33 billion, which reflects a CAGR of 17.33%.
So, considering the latest news and big jump, NMRD stock is one of the other top stock to watch for the month of February.
Sharps Technology, Inc. (NASDAQ: STSS) Has An Accelerated Near-Term Path Product For The Commercialization & Accelerated Revenue Growth!
With An Accelerated Near-Term Path For Product Commercialization And Expanded Manufacturing Capacity, Sharps Technology, Inc. (NASDAQ: STSS) Will Have The Ability To Support The Industry With Innovative, Market Leading Injectable Drug Delivery Solutions And Accelerate A Path Towards Revenue Growth.

Newly NASDAQ-traded Sharps Technology, Inc. (NASDAQ: STSS) offers innovative injectable syringe solutions to a global healthcare crisis that is not being talked about nearly enough.
- Most syringes waste a significant amount of injectable medicine which is thrown away with the used needle/syringe.
- This adds cost and reduces the availability of life-saving injectable drugs for us all. Whether we are looking at a pandemic response, hard-to-manufacture drug products, or managing long-term chronic illness, we can’t afford to waste this precious supply.
- The problem is sufficiently worrisome that the Federal Government is passing new legislation (January 1, 2023) requiring pharmaceutical companies to pay financial penalties for injectable drugs which are thrown away with the syringe and not injected into the patient.
Sharps Technology Company Summary
Company Name: Sharps Technology, Inc.
Ticker: STSS
Exchange: NASDAQ
Website: www.sharpstechnology.com
Sharps Technology Company Summary:
Sharps Technology is a medical device and pharmaceutical packaging company specializing in the development and manufacturing of innovative drug delivery systems.
The company’s product lines focus on low waste and ultra-low waste syringe technologies that incorporate both passive and active safety features.
These features protect front line healthcare workers from life-threatening needle stick injuries and protect the public from needle re-use.
Sharps Technology has extensive expertise in specialized prefilled syringe systems and ready to use processing.
The company has a manufacturing facility in Hungary and has partnered with Nephron Pharmaceuticals to expand its manufacturing capacity in the US.


Sharps Technology has agreed to manufactured in collaboration with Nephron Pharmaceuticals at the Inject EZ facility in West Columbia, South Carolina.
Jan. 10, 2023
Announces the advancement of the Company’s specialized prefillable syringe (“PFS”) system product line, which will be manufactured in collaboration with Nephron Pharmaceuticals at the Inject EZ facility in West Columbia, South Carolina.
Braden Miller, Sharps Director of Product Management, commented, “Sharps has developed an alternative high-quality solution to glass syringes through the use of inert polymers such as Cyclic Olefin Polymer (COP) and Cyclic Olefin Copolymer (COC), which offers a high-quality solution compared to traditional glass syringe systems.
These polymer syringes have many of the same characteristics as current pharmaceutical glass designs to support long term drug stability and increase shelf life for customers in the pharmaceutical segment. Polymer syringes can also be made into custom configurations, which can eliminate breakage, minimize dead space, reduce contamination, and support the development of custom devices including autoinjectors.
The ability to produce these innovative products using advanced manufacturing techniques creates additional advantages in the areas of quality, performance and safety when compared to similar glass syringe products. We look forward to introducing this line of next generation products to the market.”
Sharps Technology commenced manufacturing of their much-needed ultra-low waste smart safety syringe products in their European operation in Hungary.
Dec. 20, 2022
Announced they have commenced manufacturing of their much-needed ultra-low waste smart safety syringe products in their European operation in Hungary.
The plant has been producing products and will begin shipments to support the distribution and sales agreement with Nephron Pharmaceuticals by the end of the year, and customer agreements in Europe in early 2023. The production of these specialty syringe products will ramp up over the next several months to increase supply.
Sharps innovative syringe designs provide a beneficial set of features and advantages for the healthcare industry.
These syringe product features include a combination of ultra-low waste, passive and active safety, and reuse prevention.
By combining all these features and building them into a portfolio of syringe offerings, it will create product platforms that can help drive down the cost of healthcare treatments.
Sharps Technology signed of a distribution agreement with partner Nephron Pharmaceuticals
Dec. 13, 2022
Announced the signing of a distribution agreement with partner Nephron Pharmaceuticals. This is a strategic first step in building the larger partnership between the two companies and is in support of their recently announced collaboration.
“This distribution agreement opens so many possibilities for Sharps Technology and Nephron Pharmaceuticals,” commented Robert Hayes, Sharps Technology CEO. “The timing is perfect in that we are expanding our ability to supply innovative drug delivery systems at a point when the market is in demand for them. Through this distribution plan, Sharps Technology will be able to deliver increased capacity, driving growth for one of the high value product segments of our business.”

- Each year almost 20 billion injections are administered, globally and the World Health Organization (WHO) are advocating for the use of low waste syringes, with passive safety devices which engage automatically and have auto disable features.
- STSS anticipates signing its first product orders any moment now this development will represent a huge milestone for this recently debuted NASDAQ company, and should provide them with a solid foundation for growth.
- The company has raised $16M to scale operations in the coming quarters as they begin to meet the strong and growing demand for smart safety syringes
The global vaccines market is projected to grow from $61.04 billion in 2021 to $125.49 billion in 2028 at a CAGR of 10.8% in the forecast period, 2021-2028.
Vaccines are the most powerful and cost-effective way to protect billions of people in the world, and according to the WHO, immunization awareness and government initiatives have helped prevent 2-3 million deaths a year.
The Covid-19 outbreak led to a shutdown of syringe manufacturing which in turn led to a supply shortage at a global scale.
Mergers and acquisitions of companies such as STSS are highly attractive and are providing the needed innovation to the vaccine market.
The rise in the need for vaccinations and immunizations, plus a surge in the geriatric population, an increase in the number of surgical procedures, as well as a rise in chronic diseases have contributed to a demand for syringes.
Specialty Syringes – Vial Application
- Global Smart Syringe Market @ $14 billion USD by 2026 w10.0%+ CAGR
- Types: Auto-Disable Syringes and Safety Syringes
- Applications: Vaccination and Drug Delivery
- End Users: Hospitals, HMOs and Clinics
- Target Markets: North America, Europe, and ROW
- Sharps Technology listed as a supplier
The Pre-Filled Syringe (PFS)/Ready- To- Fill (RTF) syringe product segment will be a priority for the company through its collaboration with Nephron, and is expected to be a gamechanger for the company.
PRE-FILLED SYRINGES MARKET
- Pre-filled syringes have emerged as one of the fastest-growing choices for unit-dose medication as the pharmaceutical industry seeks new and more convenient drug delivery methods.
- With PFS/RTF syringes, pharmaceutical companies are able to minimize drug waste and increase product life span, while patients are able to self-administer injectable drugs at their home instead of the hospital.
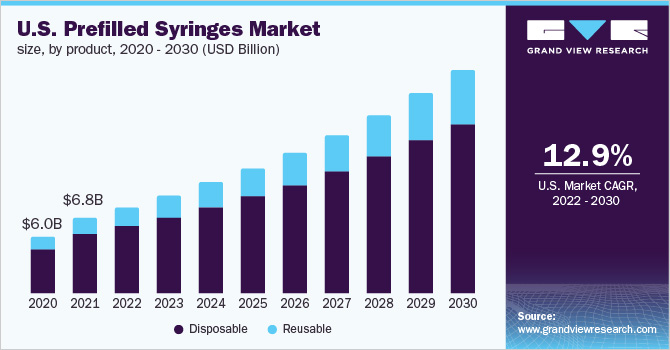
Ready-To-Use – Pre-Filled Syringes:
- Types: Plastic and Type I B/S Glass
- Applications: Vaccines, Diabetes, Complex High Value Therapies (Gene-Therapy)
- End Users: Branded Pharma, CMOs, and Biologics
- Target Markets: North America
There is a growing demand for efficient and easy-to-use drug delivery devices and increasing efforts of healthcare professionals to reduce hospital errors are the principal factors driving the market growth.

STSS announced over the summer that it had completed its acquisition of Safegard Medical’s syringe manufacturing facility in Hungary.
The manufacturing facility is located 2 hours from Budapest (160km from Budapest Airport)

- 40,000 sq. ft. factory on 250,000 sq. ft. site
- 20-year history of safety syringe manufacturing
- FDA registered since 1999
- ISO 13485 certified
- CE Mark approved products
- Injection molding and assembly expertise
- ETO on-site sterilization capacity
“The acquisition of our first manufacturing facility is an important milestone in our transformation from an R&D-focused enterprise to revenue-generating commercial operations. With the acquisition now complete, with the addition of further assembly and manufacturing capacity, our team is confident we can deliver world-class products to meet the strong and growing demand for smart safety syringes, a market forecasted to reach $14 billion globally by 2026.”
Robert Hayes, CEO of Sharps Technology

IN SUMMARY
Sharps Technology, Inc. (NASDAQ: STSS) is the newest player in the drug delivery device market to go public and could see tremendous blue-sky growth in its future as it continues to ramp up its commercialization efforts.
- The Company recently announced a significant partnership with Nephron Pharmaceuticals with an anticipated launch into the market in early 2023.
- STSS anticipates shipping first orders of vial draw product by the beginning of 2023 and ready to fill product in mid-2023 to create initial revenue in 2023 and profits in early 2024.
- With the global shortage of syringes that comply with the World Health Organization (WHO) requirements, there is a strategic opportunity for STSS to take market share and support the healthcare industry with a better drug delivery platform.
- The combination of features and benefits for the Sharps products will save lives and eliminate the waste of critically needed medical treatments and therapies for the industry!
- There’s a key opportunity for STSS to grab a big piece of the market with its proprietary smart safety syringe technology designed to eliminate two million potentially infectious accidental needlestick injuries, as well as billions of dollars in medicine wasted with today’s inefficient syringes with their low-dead space feature.
To reiterate, the company anticipates signing its first distributor agreement during the fourth quarter of 2022, which could be at any moment!
Avalon GloboCare’s (Nasdaq: ALBT) Fusion Gene Map Technology Could Be The Greatest Investment?
Literally, this could be the best investment of the year.
Why, think about this!
Avalon is establishing a leading role in the fields of cellular immunotherapy, exosome technology (ACTEX™), and regenerative therapeutics.
Why is that a big deal?
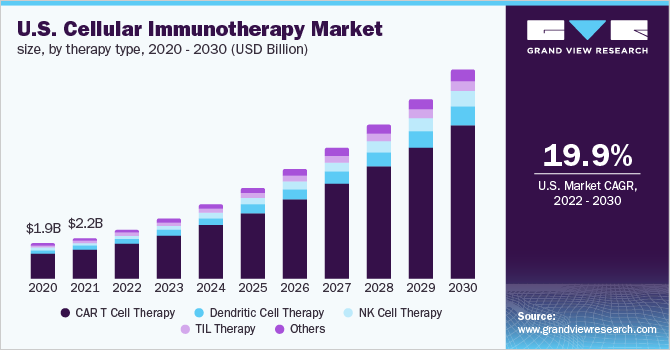
The global cellular immunotherapy size is expected to reach USD 37.97 billion by 2030!
It is estimated to register a CAGR of 22.41% during the forecast period.
This amazing growth is driven by the growing government support for innovative therapies research, rising prevalence of cancer & development of advanced cell-based immunotherapies.
Now before we get into the top 4 reasons we like ALBT, let’s do a quick summary on the company.
Avalon GloboCare Company Summary
Company Name: Avalon GloboCare Corp.
Ticker: ALBT
Exchange: NASDAQ
Website: www.avalon-globocare.com

Avalon GloboCare Company Summary:
Avalon GloboCare Corp. (NASDAQ: ALBT) is a clinical-stage biotechnology company dedicated to developing and delivering innovative, transformative cellular therapeutics, precision diagnostics, and clinical laboratory services.
Avalon also provides strategic advisory and outsourcing services to facilitate and enhance its clients’ growth and development, as well as competitiveness in healthcare and CellTech industry markets.
Through its subsidiary structure with unique integration of verticals from innovative R&D to automated bioproduction and accelerated clinical development, Avalon is establishing a leading role in the fields of cellular immunotherapy (including CAR-T/NK), exosome technology (ACTEX™), and regenerative therapeutics.


💥 RECESSION PROOF 💥
Historically, what is a great market segment to invest in during a recession, Medical!
In my opinion, The hottest vertical within The Medical market is biotech stocks.
They are safe and recession-proof.
After all, we can’t stop people from aging or from seeking treatments for a myriad of issues.
Plus, there’s growing demand for innovation in gene therapies, immune-oncology, precision medicine, machine-learning drug discovery, and treatments for unmet medical needs.
According to Grand View Research, the global biotech industry could be worth up to $2.44 trillion by 2028.
The global cellular immunotherapy market size is expected to reach
USD 37.97 billion by 2030!
It is estimated to register a CAGR of 22.41% during the forecast period.

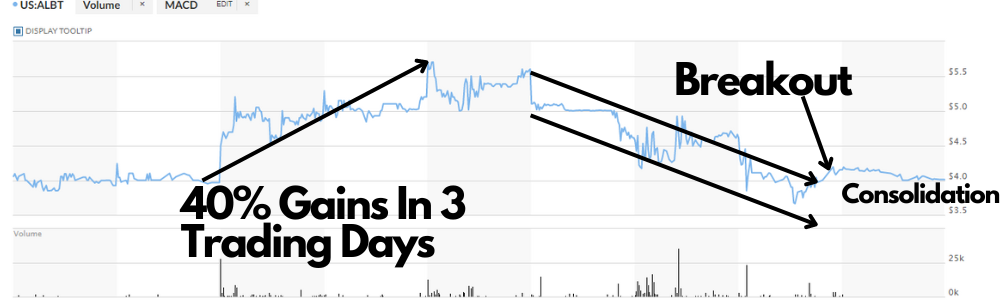
In THREE trading days this stock went up 40%!
Also, it created a base at $4 before it ran 40%!
GUESS WHAT?! It is at $4 again and appears to be ready to run!
Take a look at the buy ratings in #4 Section from Investing.com, Stock.ta and American Bulls!
3 TRUSTED sources saying, “BUY”
Now, let’s look mat what is driving these ratings and explosive gains, the amazing press released by ALBT below.

Jan. 03, 2023
Announced that it has deployed a breakthrough fusion gene map technology to be used for the goal of developing companion diagnostic kits and devices to enhance personalized clinical management of leukemia patients.
In collaboration with the Lu Daopei Institute of Hematology, a fusion gene map database from over 1,000 patients with leukemia was established and the results were previously published in the Blood Cancer Journal. Fusion genes are important genetic abnormalities in leukemia. Using advanced gene sequencing technology, called “Whole Transcriptome Sequencing” (WTS), multiple previously unknown fusion genes were identified which may potentially establish novel diagnostic and therapeutic targets.
Dec. 14, 2022
Announced that the Company completed a private placement of shares of its Series A preferred stock with the Company’s Chairman, Daniel Lu. The gross proceeds of the offering were $4.0 million, which is in addition to the previously announced private placement of $5 million of Series A preferred stock, all of which will be used to pay a portion of the purchase price for the announced acquisition of Laboratory Services MSO, LLC.
As previously announced, the Company’s Series A preferred stock is convertible into shares of the Company’s common stock at price per share equal to the greater of $1.00 or 90% of the closing price of the Company’s common stock on the Nasdaq Stock Market on the day prior to conversion. All holders of the Series A preferred stock will be restricted from selling the shares of common stock issuable upon conversion of the Series A preferred stock for a period of 9 months and will be limited to selling no more than 10% of their shares of common stock in any calendar month.
Sept. 29, 2022
The Company received a Notice of Allowance from the United States Patent and Trademark Office (USPTO) related to its QTY fusion water-soluble receptor protein platform. The patent was jointly filed with Dr. Shuguang Zhang of the Massachusetts Institute of Technology (MIT) and covers seven claims related to the technology.

ALBT has a 52-week high of $9.40, which is MORE THAN DOUBLE compared to current levels. In fact, it would be upside of 126% if the stock climbs back there.
ALBT even has a STRONG BUY Rating From Highly Respected Investing.com!

ALBT also has a BULLISH sentiment from StockTa.com right now which can be seen HERE.
Additionally, the stock has a STAY LONG rating at AmericanBulls.com which can be seen below:

Currently ALBT boasts a relatively small trading float for a NASDAQ stock at a little over $13M.
Wall Street is still uncovering this hidden gem and with a small number of shares available for trading, a sudden demand could create a major sudden upswing in price.
Now let’s review the 4 reasons we encourage you to turn your attention to ALBT.

As a quick reminder of the 4 REASONS why you should pull up ALBT right now:
- BioTech Market Is Recession Proof
- The Chart Looks Like an “Ideal Setup”
- The Press Releases are Simply Incredible
- Buy Rating Issued from Investing.com

Source 1: https://www.grandviewresearch.com/press-release/global-cellular-immunotherapy-market
Source 2: https://www.grandviewresearch.com/industry-analysis/cellular-immunotherapy-market-